U.S. Dept. of Commerce / NOAA / OAR / PMEL / Publications
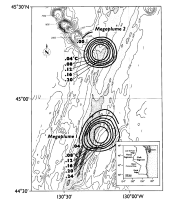
The term megaplume was coined by Baker et al. [1987] to describe a radially symmetric, 20-km-diameter plume apparently formed by a cataclysmic release of hydrothermal fluids from the axial valley of the Cleft Segment in August 1986 (Figure 2). A hydrothermal origin for this first megaplume was inferred from substantial anomalies of 3He δ3He = 117%, where δ3He = 100(R/Rair − 1); R = 3He/4He) [Lupton et al., 1989], dissolved Mn (115 nmol/kg), particulate Fe (1.16 μmol/kg), excess dissolved silica (12 μmol/kg), and temperature (0.28°C relative to ambient water of the same potential density) in the core of the plume. A cataclysmic origin (i.e., an extraordinarily large and rapid discharge) was inferred from the size and shape of the plume and the size of composition of hydrothermal minerals suspended in the plume. The megaplume was symmetric about its horizontal and vertical axes (Figures 2 and 3), implying that the plume had formed too rapidly to be elongated by local currents. Furthermore, during the 5-day sampling period the plume size and internal temperature and light attenuation structure remained unchanged, indicating that plume growth had ceased by the time of our discovery.
Figure 2. Plan view of megaplume 1 and 2 temperature anomaly (°C) contours on the 27.61σθ (~1600 m) surface. Megaplume 2 was centered about 45 km north of the original position of megaplume 1. Both plumes were radially symmetric about their vertical axis.
Figure 3. Cross sections of temperature anomaly (°C) and light attenuation (m−1) for megaplumes 1 and 2. Note that the vertical distribution of heat and particles was identical in megaplume 1 but that the particle concentration maximum was displaced downward by ~100 m in megaplume 2. Dotted lines show σθ surfaces, and sawtooth curves trace the CTD towpath. The vertical line labeled "profile" in the cross sections marks the location of the chemical profiles in Figure 5.
The unique population of hydrothermal minerals in the plume, however, required that the plume had been formed only days before we sampled it. Hydrothermal Fe oxyhydroxides, anhydrite, chalcopyrite, and amorphous silica were common in the plume. At both the center and outside edge of the plume anhydrite crystals as large as 150 μm in length were common (Figure 4). Particles of this size and shape settle at ~0.2 cm/s (170 m/d) in still water [Lerman et al., 1974], although turbulence in the plume might reduce the net speed. The presence of such large hydrothermal crystals implies a plume age of a few weeks or less, while the observation that anhydrite crystals at the edge of the plume were consistently in a more advanced state of dissolution than those at the center (Figures 4a and 4b) means that the radial expansion of the plume was not instantaneous. We deduce from these various constraints that megaplume 1 formed over no less than a few days but no longer than 2 or 3 weeks.
Figure 4. (Opposite) Scanning electron micrographs of hydrothermal minerals: (a) fresh anhydrite from the center of megaplume 1, (b) slightly degraded anhydrite from the edge of megaplume 1, (c) elemental sulfur and chalcopyrite from the center of megaplume 1, and (d) highly degraded anhydrite (overlain by other types of particles) from the center of megaplume 2. The anhydrite particles were identified by their distinctive morphology and by X ray spectra dominated by Ca and S. The arrow in Figure 4d points to the edge of the anhydrite grain.
In light of this extraordinary discovery, we gave careful attention to the detection of megaplume-type features during field work along the JDFR axis in September 1987, 13 months after the detection of megaplume 1. That attention was rewarded by the discovery of a second megaplume about 45 km north of the original megaplume location. We initially encountered megaplume 2 on September 12 during the beginning of a long CTD tow down the entire length of the Vance Segment (Figure 1). Four additional tows between September 19 and 23 (Table 1) documented its hydrographic similarity to megaplume 1 and recovered numerous water samples for chemical and mineralogic analyses.
Both megaplume 1 and megaplume 2 were centered at ~1600 m (Figure 3), but megaplume 2 had a thickness and diameter about 30% less than megaplume 1 (relative to the 0.08°C ΔT contour). The volume was ~1011 m3 inside the 0.04°C ΔT contour, or about 50% of the megaplume 1 volume. Several hydrographic sections across megaplume 2 confirmed that it was lens shaped, thus implying that it was created by a short-duration venting episode as was megaplume 1. The ΔT at the core of megaplume 2 was 0.23°C and the total excess heat
![]() (1)
(1)
where ρ is density, Cp is specific heat, and 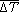 t and Vi are the mean temperature anomaly and the volume
between each ΔT contour (Figure 3), respectively, was 3.2 × 1016 J, or again about 50% of the excess
contained in megaplume 1.
t and Vi are the mean temperature anomaly and the volume
between each ΔT contour (Figure 3), respectively, was 3.2 × 1016 J, or again about 50% of the excess
contained in megaplume 1.
Both the character of the particle population and the vertical distribution of heat and particles were different in the two megaplumes. Unlike the abundant distribution of large and often uncorroded hydrothermal minerals found in megaplume 1 (Figures 4a, 4b, and 4c), only a few small and highly altered hydrothermal crystalline minerals such as barite and anhydrite were present in samples from megaplume 2 (Figure 4d). The particle mass concentration, measured by suspended matter samples and light attenuation profiles, in megaplume 2 was reduced by a factor of 4 relative to megaplume 1, presumably by the settling of particles with high fall velocities and the aggregation of very slowly settling particles. The influence of settling on the particle distribution is demonstrated by the fact that the particle maximum is ~100 m deeper than the depth of the ΔT maximum for megaplume 2 (Figure 3).
Vertical profiles of conservative and quasi-conservative hydrothermal constituents through megaplume 2 show a basic similarity to analogous megaplume 1 data. The concentrations of 3He and dissolved Mn closely follow the temperature profile (Figure 5a). The concentration profile of dissolved hydrothermal silica was calculated by subtracting from the observed profile a silica profile from near-axis stations outside the megaplumes. This profile of excess silica also matches the profile of temperature, 3He, and Mn (Figure 5a).
Figure 5. Vertical profiles of (a) dissolved and (b) particulate hydrothermal constituents of megaplumes 1 and 2. The megaplume 1 profile was taken on the plume edge, whereas the megaplume 2 profile was taken near the center (see Figure 3), so profiles of conservative constituents are similar even though megaplume 1 was actually more concentrated than megaplume 2.
Particulate species exhibit various types of profiles (Figure 5b), presumably as a function of the mean settling velocity of the dominant particle type with which each element is associated. Fe closely follows the trend of the dissolved components, because most of the Fe is in submicrometer-sized amorphous oxyhydroxide precipitates [Walker and Baker, 1988]. Such precipitates composed ~40% by weight of megaplume 1 solids [Baker et al., 1987]. Their very low settling velocity (<<1 × 10−5 cm/s as calculated by the Stokes equation) means their removal is principally by biological or physical aggregation rather than settling. The hydrothermal Fe precipitates may thus be considered quasi-conservative in the sense that their relative concentration profile is stable for many months.
Figure 5. (continued)
Concentrations of particulate S in megaplume 2 are also lower than in megaplume 1 (Figure 5b). S occurs principally in coarse-grained hydrothermal precipitates such as anhydrite, chalcopyrite, pyrite, and barite that settle rapidly and are quickly lost from the plume [Baker and Massoth, 1987; Feely et al., 1987]. Even the vertical distribution of S in megaplume 1 shows clear evidence of settling of the particulate S phase.
The vertical distribution of particulate Mn in megaplume 2 is unique among these profiles in having a substantially higher concentration than was found in megaplume 1 (Figure 5b). About 7% of the present Mn inventory of the plume (plus an unknown fraction that has presumably been lost to settling) is particulate, compared to 1% in megaplume 1. The fraction of Mn in the particulate phase was smaller in both megaplumes than that reported for plumes at 19°S on the East Pacific Rise (EPR) [Klinkhammer and Hudson, 1986] and on the Mid-Atlantic Ridge [Klinkhammer et al., 1985].
The linear relationships between 3He and other conservative and quasi-conservative constituents in both megaplumes are similar to those characteristic of high-temperature vent fluids from a variety of locations (Figure 6). The excess heat/3He ratio from both plumes is ~3 × 1017 J/mol 3He (3.2 × 1012 cal/cm3 STP), although the scatter of the data is greater in megaplume 1. This ratio is higher than vent fluids sampled at 21°N and 13°N on the EPR and at 44°40′N on the JDFR, but well below those from the Guaymas Basin (Figure 6a).
Figure 6. Scatter plots of 3He concentration versus (a) enthalpy anomaly, (b) dissolved silica anomaly, and (c) dissolved Mn in megaplume 1 and megaplume 2. Plotted for comparison are corresponding slopes (forced through a point representing ambient seawater) from high-temperature fluids sampled on the East Pacific Ridge, Guaymas Basin, and the southern Juan de Fuca Ridge at 44°40′N. The following data are given: 3He and heat data from 21°N [Welhan and Craig, 1983], 13°N [Merlivat et al., 1987], Guaymas [Lupton, 1983], and southern JDFR [Kennedy, 1985]; silica from 21°N [Von Damm et al., 1985a], Guaymas [Von Damm et al., 1985b], and southern JDFR [Von Damm and Bischoff, 1987]; Mn at 21°N [Von Damm et al., 1985a, 1983], Guaymas [Von Damm et al., 1985b], and southern JDFR [Von Damm and Bischoff, 1987].
The trend of the excess silica/3He relationship is ~2 × 109 mol/mol for the megaplumes and is an analog of the excess heat/3He relationship insofar as it is higher than the vent fluid regressions from the EPR and the JDFR but lower than the regression from the Guaymas Basin fluids (Figure 6b). The excess silica curve is less precise than either the heat or Mn curves because of the relatively low enrichment of silica in hydrothermal fluids and the consequent difficulty in determining the true nonhydrothermal level of silica in the water column. Our estimate of hydrothermal silica is a maximum value because we have not accounted for the addition of silica into the megaplume by entrainment of deep, silica-rich water. The existence of a direct relationship between silica and 3He is nevertheless unequivocal.
The trend of the dissolved Mn (>93–99% of total Mn)/3He relationship is ~2.3 × 107 mol/mol, a factor of ~3–10 less than sampled vent fluids (Figure 6c). This difference is not unexpected because sampling at the 44°40′N vent site on the JDFR [Baker and Massoth, 1986a] and at 21°N on the EPR [Lupton et al., 1980] has shown that Mn concentrations relative to heat or 3He are lower in the neutrally buoyant plume than in the sampled vent fluids. The cause of this difference remains uncertain. In fact, the megaplume Mn/3He trend is intermediate between that of plumes from 44°40′N and the 21°N plumes.
A central question is whether megaplume 2 was a separate event or merely a reobservation of megaplume 1. While there is circumstantial evidence for both views, our judgment is that the weight of evidence favors the case for separate megaplumes. Arguments for individual events are based on calculations of the expected horizontal advection velocity of the plume, on the size and mineralogy of individual particles in megaplume 2, and on measurements of the microbial binding rate of Mn in hydrothermal plumes.
Current meter records over and adjacent to the Cleft Segment [Cannon and Pashinski, 1987, also unpublished manuscript, 1989] indicate that net velocities over periods of 11 to 13 months and at depths between 1000 and 2000 m are typically 1–2 cm/s. Progressive vector diagrams of four such records (Figure 7) imply annual displacements of 200 to 600 km, a factor of 5 to 15 greater than the observed separation between megaplumes 1 and 2 (Figure 3). While the calculation of actual Lagrangian displacements of water parcels originating at the mooring sites would require a dense regional network of moorings, the large displacements inferred from these single moorings suggest that a megaplume is unlikely to be found near its origin on the ridge crest after a year's time. We also note that the magnitude of these displacements matches that found for a recently tracked "Meddy" centered at 1000 m in the eastern Atlantic [Armi et al., 1988].
Figure 7. Progressive vector diagrams from current meters deployed near the megaplume 1 site. The paths represent the cumulative displacement inferred from the net velocity of water past each mooring. The Juan de Fuca Ridge axis is shown for scale. Mooring V1 was recorded from May 25, 1984, to June 17, 1985; V8, from June 9, 1985, to April 13, 1986; and V9, from August 16, 1986, to July 27, 1987. Crosses represent 30-day intervals along each path. Circles on the ridge crest show the locations of megaplumes 1 and 2.
We can also document a minimum displacement velocity for megaplume 1 of ~0.2 km/d from the fact that CTD taken at the location of the plume center 55 days after its initial discovery showed no trace of the plume, indicating that the plume moved a distance of at least 1 radius (~10 km) in that time.
Mineralogical samples available from megaplume 2 provide additional evidence for a plume age of less than 13 months. The anhydrite particle shown in Figure 4d appears to be part of an aggregate with an estimated settling rate of ~0.01 cm/s (9 m/d), fast enough to have settled out of the plume in much less than a year unless substantial vertical currents persist in the plume throughout its life. Furthermore, the laboratory and field experiments of Feely et al. [1987] show that anhydrite dissolves rapidly in seawater, so large crystals are unlikely to have survived for 13 months.
Finally, Cowen et al. [1988] estimated the age of both megaplume by calculating the removal rate of dissolved Mn onto particles as determined by in situ measured microbial Mn binding rates. Estimated ages were 1–3 days for megaplume 1 and about a month for megaplume 2 (assuming that the original concentration of dissolved Mn in megaplume 2 was the same as in megaplume 1).
The principal argument against separate events is the similarity of the ratios of excess heat, excess dissolved silica, and dissolved Mn to 3He in both plumes (Figure 6). If a similar process created both plumes, however, a compositional similarity would not be unexpected. Campbell et al. [1988] and Bowers et al. [1988] observed that the composition of fluids discharging from individual vent fields on the EPR was uniform over a several-year sampling interval. They argued that this constancy was maintained through buffering of the hydrothermal fluids by the host rock; if both megaplumes originated from same or petrographically similar reservoirs, they should be chemically similar.
While the evidence does not conclusively prove that megaplumes 1 and 2 were separate events, it is suggestive of it and consistent with the hypothesis that megaplume 2 formed in the vicinity of the megaplume 1 location and underwent net northward advection at ~1 cm/s for a few months before its discovery in September 1987. Although we assume separate events in the following discussions, none of the conclusions we reach are dependent upon that assumption.
Go back to previous section or forward to next section