
 )
is calculated as
)
is calculated as U.S. Dept. of Commerce / NOAA / OAR / PMEL / Publications
Hydrothermal plumes form above sites of venting because of the buoyancy of the hot hydrothermal fluids that rise, entraining ambient sea water with a consequent continuous increase in plume volume, until neutral buoyancy is achieved and the plume disperses laterally. Models describing the physical and chemical properties of hydrothermal plumes have been presented by several workers [e.g., Middleton and Thomson, 1986; Little et al., 1987; Speer and Rona, 1989; McDougall, 1990; Rudnicki and Elderfield, 1992; Lavelle, 1994], all based upon the principles for turbulent entrainment in plumes described by Morton et al. [1956] and Turner [1973]. Using these models it is possible to calculate heat fluxes from plume and local hydrographic data, and to predict the ratio of entrained seawater to hydrothermal fluid as a function of plume height. During plume rise, ambient seawater is entrained from a range of depths such that the concentration of a property in the neutrally buoyant plume is a function of both the concentration and flux of that property in the vent fluid, and of the background profile of the property integrated over the whole height of rise of the buoyant plume. These complexities are discussed more fully in the following sections, and especially in accompanying papers by McDuff [this volume], Lupton [this volume], and Helfrich and Speer [this volume].
Quantification of hydrothermal temperature anomalies is complicated by the
variable background profiles of temperature and salinity found in the world
ocean. In the deep Pacific, the ambient salinity gradient increases with depth
and so the neutrally buoyant plume is warmer and saltier than its surroundings.
An estimate of the hydrothermal temperature anomaly (
 )
is calculated as
)
is calculated as
![]() (1)
(1)
where k and b are the slope and intercept, respectively, of the
generally linear relationship between potential temperature ( )
and potential density (
)
and potential density (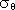 ) in hydrothermally
unaffected water immediately above the neutrally buoyant plume. To properly
compare this
) in hydrothermally
unaffected water immediately above the neutrally buoyant plume. To properly
compare this  value in the neutrally buoyant
plume with the hydrothermal heat input at the seafloor source, however, the
plume values must be corrected for the effect of local hydrography and vent
fluid salinity.
value in the neutrally buoyant
plume with the hydrothermal heat input at the seafloor source, however, the
plume values must be corrected for the effect of local hydrography and vent
fluid salinity.
In the Atlantic and wherever else the salinity gradient is negative above the ridge crest, the neutrally buoyant plume is cooler and fresher than the surrounding seawater. Equation (1) gives a negative value in this case, and hydrographic mapping of the plume is problematic at best. Evaluation of temperature anomalies must be considered within the framework of a plume model [e.g., Speer and Rona, 1989; McDougall, 1990; Rudnicki and Elderfield, 1992]. Thus most plume mapping in the Atlantic has depended on optical or chemical indices of hydrothermal discharge.
Both light attenuation and light scattering have been widely used to define hydrothermal plumes. Unlike conservative hydrographic tracers, optical tracers are nonconservative and depend on an ever-changing balance between particle production by precipitation and biological growth, and particle loss by deposition, dissolution, and ingestion. Hydrothermal fluids with a very low level of precipitable species, such as the vapor phase of a phase-separated fluid, or a fluid discharging through a thick layer of sediments, may yield a plume with a tenuous optical signal, but optically invisible plumes are rare. The only documented case we are aware of is a CH4 plume with no nephelometer or total dissolved Mn signal near 15°N on the Mid-Atlantic Ridge (MAR), a plume hypothesized to arise from fluid circulation in ultrabasic (low silica) rocks rather than basalt [Charlou et al., 1991a]. Moreover, optical tracers are often more sensitive and simpler to interpret than hydrographic tracers, since their background profiles are roughly vertically uniform. Their nonconservative nature can add to their usefulness, since an optical signal decreases more sharply than temperature with distance from its source.
Hydrothermal fluids are enriched by up to a factor of 107 in several key tracers (e.g., Mn, Fe, CH4, H2, 3He, and many other trace species) relative to typical oceanic deep waters, so chemical anomalies associated with hydrothermal plumes can often be detected at significant distances away from hydrothermal vent sites. (The chemistry of hydrothermal fluids and plumes is more fully discussed by Lilley et al. [this volume], Lupton [this volume], Kadko et al. [this volume], and Von Damm [this volume].) Dissolved manganese (Mn) and methane (CH4) are commonly used to define hydrothermal plumes for three reasons. First, both are enriched ~106-fold in high-temperature vent fluids relative to ambient seawater [e.g., Welhan and Craig, 1983; Von Damm, 1990], so their concentrations in neutrally buoyant hydrothermal plumes directly above hydrothermal vent sites are ~102-fold higher than typical oceanic deep water. Second, both can be analyzed precisely at sea. Third, the contrasting quasiconservative behavior of Mn and distinctly nonconservative behavior of CH4 provide dynamical information about plumes. Because neutrally buoyant plumes overlie a much greater area of the midocean ridge crest than is occupied by active hydrothermal chimneys and mounds, these water column enrichments present geochemists a magnifying glass with which to prospect for new hydrothermal vent sites.
Fig. 1. Schematic comparison of three methods of shipboard plume mapping and sampling.
Hydrographic and optical measurements are commonly combined in the form of
a conductivity/temperature/depth (CTD) system interfaced with a transmissometer
or nephelometer (or both). A basic sampling strategy is simply vertical casts
widely spaced along the ridge axis (Figure 1).
Because hydrothermal plumes are dynamic features with steep horizontal and vertical
gradients, however, it is useful to adopt a sampling scheme that will reduce
the problem of temporal and spatial aliasing as much as possible. Data return
should be maximized from the plume horizon and minimized from waters above the
plume where little useful information is present. An efficient strategy in mapping
and sampling neutrally buoyant plumes is thus to conduct lengthy "tow-yos,"
continual raisings and lowerings of the instrumentation through the plume while
slowly steaming (Figure 1). Experience shows
that a typical CTD package can be towed with standard electromechanical cable
at a rate of about
The majority of geochemical studies associated with hydrothermal plume surveys up to about 1990, and which continue in widespread use today, required conventional collection of seawater samples using CTD-rosette systems followed by ship-board or shore-based analyses for geochemical tracers. To accommodate the enlarged data sets required to address more complex geochemical problems, much investment has been made recently in novel instrumentation which can achieve greater sampling coverage or even produce real-time in situ geochemical analyses. This instrumentation will generate data in a form comparable to that readily obtainable from hydrographic and optical sensors, thus facilitating improved understanding of the biogeochemical processes active in plumes.
The first advance in geochemical studies of hydrothermal plumes came from the
IFREMER laboratory's development of a continuous underway towed water sampler--the
"Palanquée Dynamique" or "Dynamic Hydrocast" [Bougault
et al., 1990]. This system comprises four sets of water sampling rosettes,
each with a CTD sensor package and data logger, that are clamped to a cable
and towed at different, known heights above the seafloor (Figure
1). Each set of water sampling rosettes comprises ten water-sampling bottles
which fill progressively, and sequentially, as the rosette package is towed
through the water column. The gearing on the rosettes can be adjusted so that
each bottle fills progressively over
Although the Dynamic Hydrocast system enables systematic and continuous water-sampling,
only a limited number of samples can be collected and these must be analyzed
onboard or ashore. An alternative approach was taken by Johnson and co-workers
at the Moss Landing Marine Laboratories. They developed a novel instrument package,
the Submersible Chemical Analyzer (SCANNER), that could
carry out analyses of nutrients, sulfide, Mn and Fe in situ [Johnson
et al., 1986a,b;
Coale
et al., 1991; Chin
et al., 1994]. The system is a deep-towed spectrophotometer that measures
dissolved Mn and total dissolved Fe (Fe(II) and Fe(III)) every 5 seconds in
seawater that has previously been passed through a 10 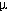 m
mesh prefilter. The system connects routinely to a conventional CTD package
augmented with a nephelometer and/or transmissometer. Coale
et al. [1991] and Chin
et al. [1994] used the SCANNER to map dissolved Fe and Mn anomalies
in the range
m
mesh prefilter. The system connects routinely to a conventional CTD package
augmented with a nephelometer and/or transmissometer. Coale
et al. [1991] and Chin
et al. [1994] used the SCANNER to map dissolved Fe and Mn anomalies
in the range
More recently, SUAVE (Submersible System Used to Assess
Vented Emissions), an enhanced version of the SCANNER system,
has been developed by NOAA's Pacific Marine Environmental Laboratory. The SUAVE
system comprises a total of six on-line colorimetric chemical detectors for
Mn, Fe(II), Fe(II+III), Si, H2S, and one of PO4 or Cl,
currently under development for plume and diffuse-flow studies, respectively
[Massoth
et al., 1991] (G. J. Massoth, personal communication, 1994). The system
can currently be deployed for at least
Sensitivity of both the SCANNER and SUAVE systems are limited by the colorimetric
techniques available for dissolved Mn and Fe analyses, originally
To overcome this potential problem, Klinkhammer
[1994] has developed an even more sensitive dissolved Mn analyzer, ZAPS
(Zero Angle Photon Spectrophotometer), that can detect dissolved Mn to ambient
seawater concentrations 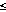 1 nmol l-1),
1 nmol l-1),
Return to previous section or go to next section